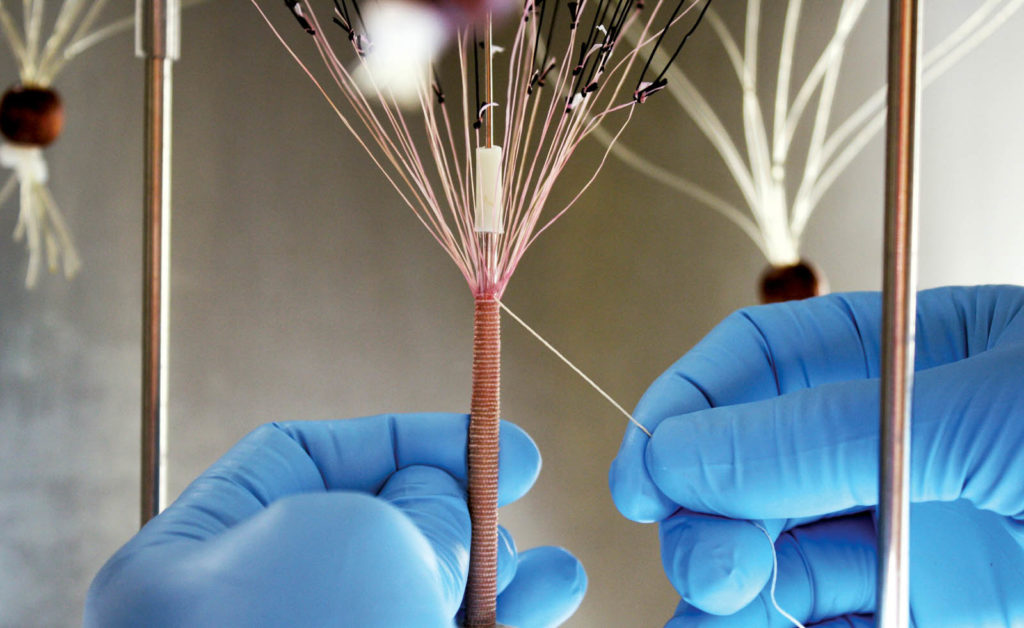
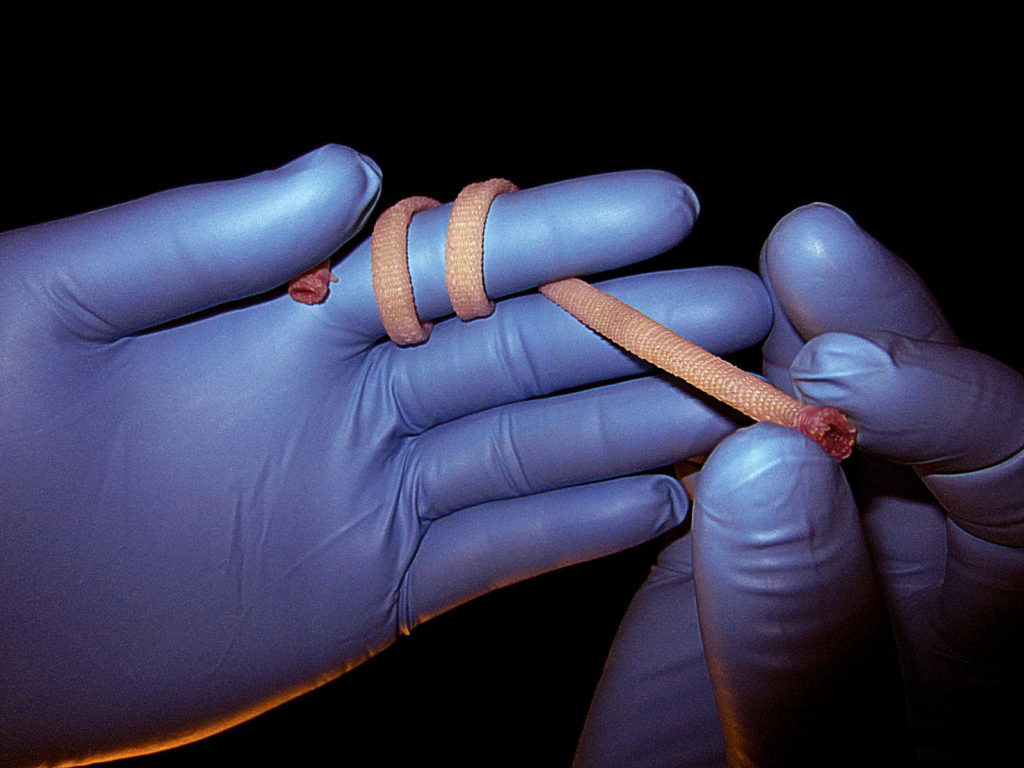
New bio yarn shows breakthrough possibilities
Researchers working at the University of Bordeaux/INSERM have created yarns of human cells from sheets of cell-assembled extracellular matrix, or CAM. The findings, reported in www.materialstoday.com, are a significant achievement because the CAM can be mass-produced for clinical applications including tissue engineering. However, the process is complex and the usefulness of sheets for creating different shapes is limited. “We make yarn by cutting the CAM sheet into ‘ribbons’ that can be used directly or twisted into ‘threads’ [with] slightly different mechanical properties,” says Nicolas L’Heureux, University of Bordeaux/INSERM, who led the research. The yarns, which can be 2-10 mm wide and up to 3 m long, can be used as they are as simple sutures for wound repair or woven, knitted or braided into almost any shape. Multiple yarns can be twisted together to create thicker, stronger yarns if different mechanical properties are needed. The yarn can be simply dried, stored frozen and rehydrated when needed, the researchers report.
Machine converts to knitting masks
Karl Mayer, a global warp-knitting machine manufacturer based in Obertshausen, Germany, says that its tricot machines with two guide bars for elastic fabrics can be converted to the production of tapes for masks. The company estimates enough tape could be produced for up to 15 million masks per month and that European machines could be put to work to augment Asian production. It is also developing protective masks, as well as instructions for their high-speed production. The masks developed at Karl Mayer are manufactured in one piece, without sewing. Photo: Karl Mayer.

New durable water repellent
Tokyo-based Toray Industries Inc. has introduced Kudos™ XT, a new durable water repellent (DWR) delivering higher levels of durability for both abrasion and machine washing. The breakdown of DWR treatment and subsequent “wetting out” of a garment’s outer layer has been an issue since the advent of waterproof/breathable fabrics. DWR treatments improved over time, but consumers objected to the new levels of toxicity, culminating with C8 DWR being banned in many markets. Kudos XT is available in a perfluorocarbon (PFC)-free version, as well as C6, a level less toxic than C8. Each version prevents an outer layer from wetting out, even under heavy rain conditions. The company is targeting diverse markets including outdoor, snowsports, athleisure, sportswear and work uniforms. Photo: Toray Industries.

Company designs firefighting gear for women
After five years of research and development, a women-owned company is ready to launch a line of female-specific uniforms for wildland firefighters. Korena Hallam and Summer Hurd, founders of Green Buffalow of Fort Collins, Colo., say the company’s products address problems with conventional uniforms made for what is largely a male-dominated field: there is no size chart in the standard for female upper torso garments; lower torso female sizing is almost a mirror image of the men’s lower torso size table; restrictive design requirements result in ill-fitting garments; and due to poor fit, some individuals experience chafing and bleeding. The company has partnered with industry experts to design uniforms that will reduce heat stress and allow optimal movement. Photo: Green Buffalow.

FDA grants approval for AR Tech’s isolation unit
AR Tech, Fontana, Calif., has received emergency approval for the use of its Patient Isolation Transportation Unit (PITU) from the U.S. Food and Drug Administration (FDA). The unit can be used by health-care providers to temporarily isolate and transport patients with a confirmed or suspected COVID-19 infection. The PITU protects medical professionals, patients and visiting family members against respiratory droplets and infection by using a negative pressure enclosure that surrounds the patient. Patients can be treated by medical staff, and family members who are properly attired can visit. Dr. Mark Comunale, chief medical officer for patient safety, Arrowhead Regional Medical Center, Colton, Calif., worked on developing the unit in 2015 after the Ebola outbreak. AR Tech joined Comunale’s efforts two years ago. Photo: AR Tech.

 TEXTILES.ORG
TEXTILES.ORG


